Case Study: An 8-Year-Old Female with Turner Syndrome Presents with Abdominal Pain and Bloody Stools
Sayeeda M. Kamal,Rummanu Yeasin
Smart Medical Care PC, Jamaica NY
Corresponding Authors: Maksud Chowdhury MD
Correspondence: Smart Medical Care PC. Jamaica NYTurner syndrome (TS), a female-only condition, is one of the most common chromosomal abnormalities worldwide. The most common presenting clinical features of TS are short stature and gonadal dysgenesis. TS is frequently associated with autoimmune conditions including thyroid disease, diabetes mellitus, Celiac disease, and inflammatory bowel disease (IBD). Several studies suggest an increased incidence of IBD in patients with TS. Here we present a case of TS who was diagnosed with Crohn’s disease at the age of 9 years. The patient was on growth hormone treatment for growth retardation. She developed bloody diarrhea and iron deficiency anemia, which subsequently led to the diagnosis of Crohn’s disease. Systematic evaluation of patients with TS, recognition of the association with IBD, and early intervention may improve quality of life, prevent future complications, and may lengthen life expectancy in these patients.
Case Study: An 8-Year-Old Female with Turner Syndrome Presents with Abdominal Pain and Bloody Stools
Sayeeda M. Kamal,Rummanu Yeasin
Smart Medical Care PC, Jamaica NY
Corresponding Authors: Maksud Chowdhury MD
Correspondence: Smart Medical Care PC. Jamaica NY
Introduction
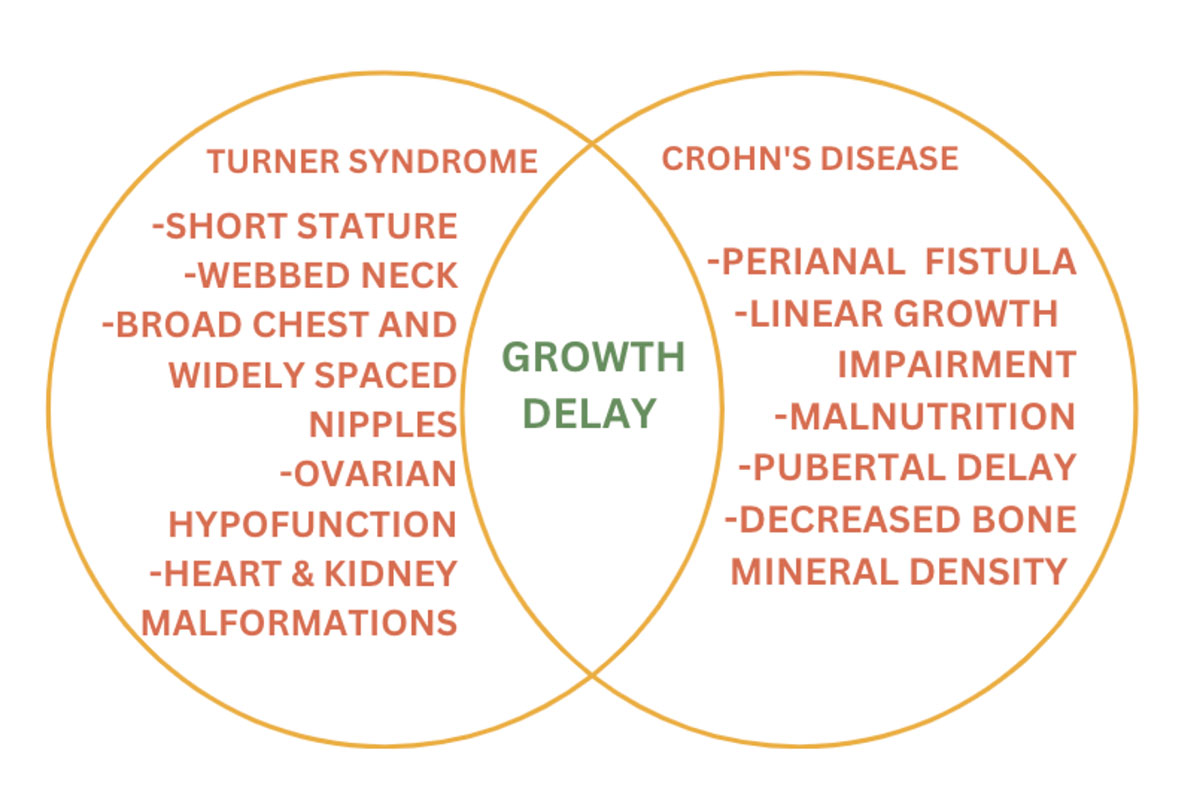
Turner syndrome is a clinical syndrome that arises in females due to the complete or partial absence of the X chromosome. 45XO is a classical karyotype of TS. A TS patient may also present with a mosaic pattern with some of the cells having the classical karyotype and others having an absent or abnormal X chromosome. The risk of autoimmunity in TS is well recognized and the risk is approximately twice as high as in the general female population.1
Inflammatory bowel disease (IBD) comprises two chronic conditions Crohn’s disease (CD) and Ulcerative colitis (UC). The association between IBD and TS has been well established. Colitis in patients with TS is seen to have a more progressive course than in patients with colitis alone.2 Here we report a case of TS, who developed Crohn’s disease.
Case Presentation
An 8-year-old female born via normal vaginal delivery at full-term gestation had uncomplicated prenatal and postnatal courses. She was referred to an endocrinology clinic at the age of 8 years for evaluation of short stature, obesity, and prediabetes. Her height was 47 in, weight was 76 lbs., BMI was 24.2 and HBA1c was 5.9%. At the endocrinology clinic, the patient was suspected to have growth failure due to TS. Prediabetes was detected. No thyroid dysfunction or celiac disease was detected. Cytogenetic analysis revealed a mosaic pattern of 46X,del(X)(p11.2)[12]/45,X[8]. The patient started growth therapy at the age of 8 years. She subsequently developed bloody stool and vomiting and was evaluated in the GI clinic post ER visit. She was recognized to have iron deficiency anemia. Based on all her physical and clinical findings IBD was suspected and a colonoscopy and biopsy were done.
.jpg)
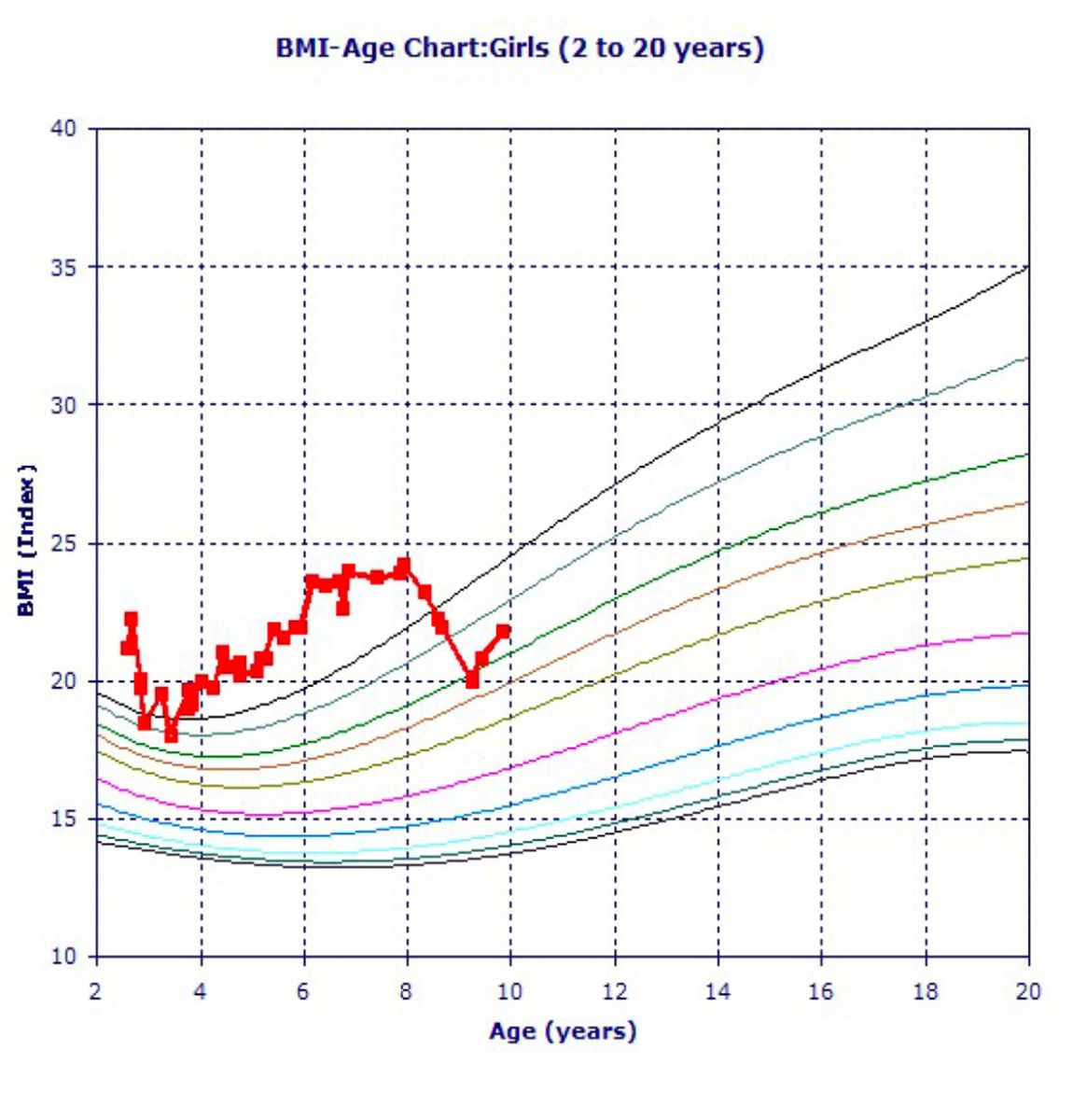
Figure 1: Our patient’s BMI-Age Chart
Figure 2: Our patient’s Stature-Age Chart
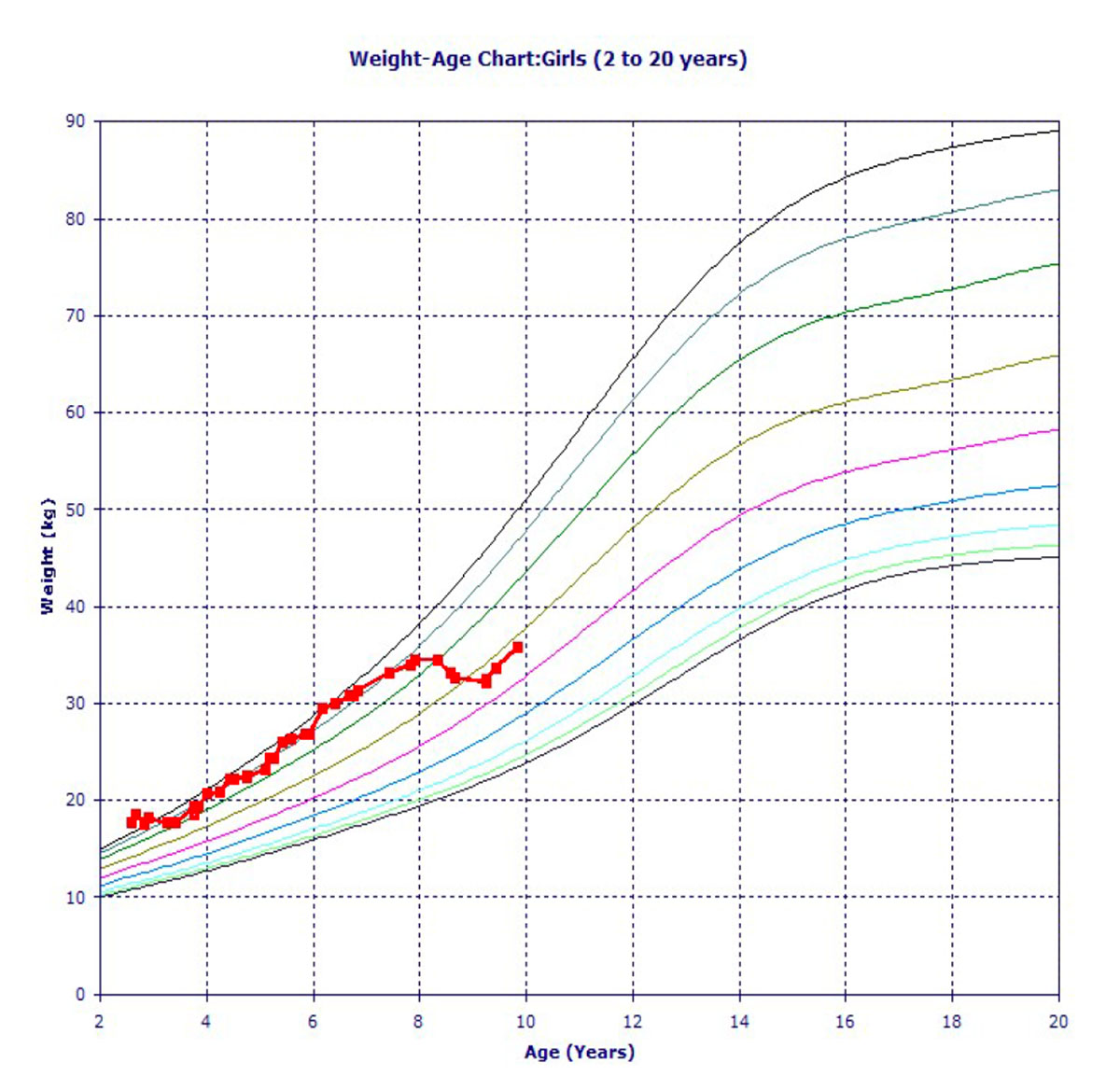
Figure 3: Our patient’s Weight-Age Chart
History of Present Illness: An 8-year-old female with recurrent bloody stools and a history of anemia presents to ER for possible appendicitis. Imaging did not show any inflammation of the appendix consistent with appendicitis, and the patient is referred to outpatient GI to undergo evaluation for possible inflammatory bowel disease (IBD).
Social History: Only child, lives with both parents, no one smokes at home.
Allergies: NKDA
Past Medical History: Turner syndrome
Past Surgical History: None
Medications: growth hormone
Family History: No known family history
Physical Exam
Vitals: Temp: 97.1 F, Height: 47 in, Weight: 76 lbs., BMI: 24.2, BP: 110/75, SpO2: 100%
Review of Symptoms:
Constitutional: short stature
Skin: negative
Breast: negative
Eyes: negative
ENMT: negative
Respiratory: negative
Gastrointestinal: bloody stool
Genitourinary: vulvar vitiligo
Musculoskeletal: negative
Hematologic/Lymphatic: anemia
Psychiatric: negative
Initial Evaluation
Laboratory Studies
Cytogenetic analysis revealed sex chromosome mosaicism. Eight of 20 metaphases had a 45,X karyotype. The remaining cells had a normal X chromosome and a second X chromosome with a deletion of most of its short arm. IGF-1 115, IGFBP-3 3.35, Hemoglobin A1c 5.8, T4 1.81, TSH 4.560, cortisol 21, Celiac disease panel negative
MRI of abdomen and pelvis shows: liver normal, spleen normal, pancreas normal, gallbladder/biliary tree normal, adrenals normal, kidney normal, lymphadenopathy/retroperitoneum no adenopathy, bowel: stomach normal, proximal small bowel normal, terminal ileum normal, colon/rectum: allowing for the degree of distention, there is mild circumferential wall thickening of the transverse colon, descending colon, sigmoid colon, and rectum with associated transmural hyperenhancement.
Colonoscopy revealed erythematous, friable (inflamed and ulcerated mucosa in the descending colon at the splenic flexure and in the distal transverse colon).
Biopsy revealed active inflammation involving the duodenal bulb, stomach, and colon. The inflammation is most pronounced from the distal transverse to the sigmoid colon where it is characterized by multifocal acute cryptitis, neutrophilic infiltration of the surface epithelium, and increased neutrophils throughout the lamina propria. Features of chronicity including increased to full thickness lamina propria, lymphoplasmacytosis and glandular architectural distortion are also noted within this region. A single non-caseating epithelioid granuloma is identified within the cecum. The histological features are compatible with inflammatory bowel disease, favor of Crohn’s disease, provided other etiologies including infection and drug-induced injury/inflammation have been excluded. Based on these findings, a diagnosis of Crohn’s disease was made.
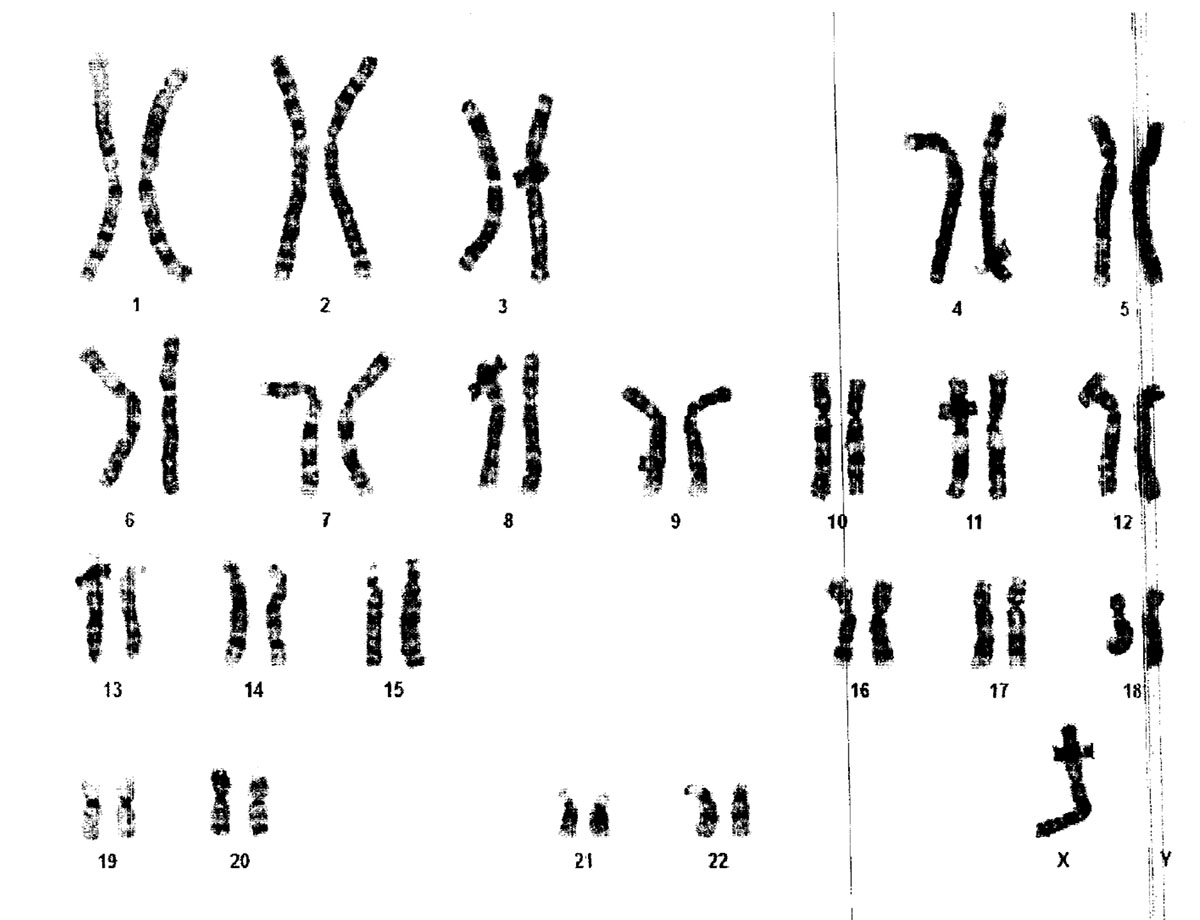
Figure 4: Cytogenetic result of our patient, taken from blood sample
Differential Diagnosis
Crohn’s Disease
Infection
Drug induced injury
Diagnosis
Based on the gastric MRI and biopsies, a diagnosis of Crohn’s disease was made. Drug induced injury ruled out because patient was only ever on growth hormone and the occasional over the counter Tylenol and Motrin.
Management
After the initial diagnosis of Turner syndrome was made, the patient was sent for cardiac evaluation. It was notable for the slight prolongation of the corrected QT interval on her EKG. Her cardiac evaluation was otherwise normal, and there was no evidence of coarctation of the aorta or congenital heart disease. Recommendations include avoiding medication that prolongs QT interval (i.e., macrolide antibiotics, Benadryl), and reassessment in 2 months.
Patient was also seen in hematology and ruled out for celiac disease.
As our patient’s BMI was on the higher side, and her A1c was increased, it was advised that she control her diet. Hence after age 8, after diet, exercise, and lifestyle modifications, her BMI and weight-age curves were coming down.
Discussion
It is important to start early treatment and intervention as soon as the diagnosis of Turner syndrome is made. Many genetic syndromes share relevant features with Crohn’s, such as poor growth or short stature, and these similar symptoms can delay the diagnosis of IBD6. There is also a recent increase in diagnosing CD in patients with Turner syndrome and Down syndrome6. It is also important to realize that a patient can be more susceptible to developing drug toxicities, infections and surgical complications due to the presence of other comorbidities in genetic conditions. As in our patient, it was recommended to avoid medication that prolongs QT interval. Because our patient has mild CD, she does not need any medication at the moment but will require close follow up to make sure the disease does not progress. Based on moderate to severe CD, medical therapy includes medication, ileocolic resection, and dietary therapy.
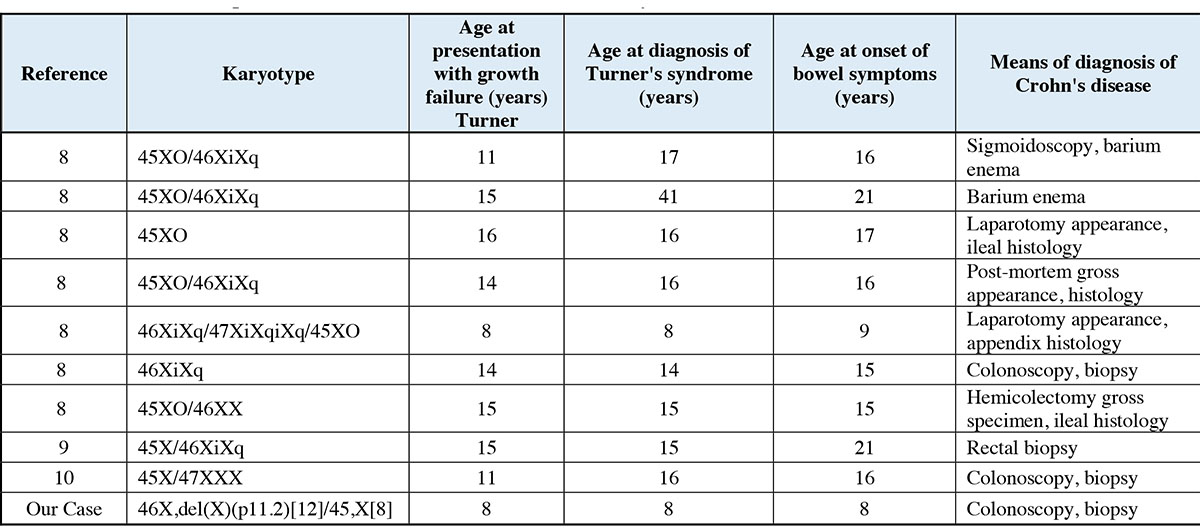
Figure 6: Summary of documented cases of concomitant Turner syndrome and Crohn’s disease
Human Ethics
Maternal consent was received on behalf of the patient for this paper. Written informed consent was obtained from the mother of the individual for the publication of any potentially identifiable images or data included in this article.
The authors have declared that no competing interests exist.
Funding
The authors received no specific funding for this work
- De Sanctis V, Khater D. Autoimmune diseases in Turner syndrome: an overview. Acta Biomed. 2019 Sep 6;90(3):341-344. doi: 10.23750/abm.v90i3.8737. PMID: 31580326; PMCID: PMC7233727
- Durusu M, Gürlek A, Simsek H, Balaban Y, Tatar G Coincidence or causality : celiac and Crohn diseases in a case of Turner syndrome. Am J Med Sci. 2005 Apr;329(4):214-10.1097/00000441-200504000-00010. PMID: 15832107.
- Sazonovs A, Stevens CR, Venkataraman GR, Yuan K, Avila B, Abreu MT, Ahmad T, Allez M, Ananthakrishnan AN, Atzmon G, Baras A, Barrett JC, Barzilai N, Beaugerie L, Beecham A, Bernstein CN, Bitton A, Bokemeyer B, Chan A, Chung D, Cleynen I, Cosnes J, Cutler DJ, Daly A, Damas OM, Datta LW, Dawany N, Devoto M, Dodge S, Ellinghaus E, Fachal L, Farkkila M, Faubion W, Ferreira M, Franchimont D, Gabriel SB, Ge T, Georges M, Gettler K, Giri M, Glaser B, Goerg S, Goyette P, Graham D, Hämäläinen E, Haritunians T, Heap GA, Hiltunen M, Hoeppner M, Horowitz JE, Irving P, Iyer V, Jalas C, Kelsen J, Khalili H, Kirschner BS, Kontula K, Koskela JT, Kugathasan S, Kupcinskas J, Lamb CA, Laudes M, Lévesque C, Levine AP, Lewis JD, Liefferinckx C, Loescher BS, Louis E, Mansfield J, May S, McCauley JL, Mengesha E, Mni M, Moayyedi P, Moran CJ, Newberry RD, O'Charoen S, Okou DT, Oldenburg B, Ostrer H, Palotie A, Paquette J, Pekow J, Peter I, Pierik MJ, Ponsioen CY, Pontikos N, Prescott N, Pulver AE, Rahmouni S, Rice DL, Saavalainen P, Sands B, Sartor RB, Schiff ER, Schreiber S, Schumm LP, Segal AW, Seksik P, Shawky R, Sheikh SZ, Silverberg MS, Simmons A, Skeiceviciene J, Sokol H, Solomonson M, Somineni H, Sun D, Targan S, Turner D, Uhlig HH, van der Meulen AE, Vermeire S, Verstockt S, Voskuil MD, Winter HS, Young J; Belgium IBD Consortium; Cedars-Sinai IBD; International IBD Genetics Consortium; NIDDK IBD Genetics Consortium; NIHR IBD BioResource; Regeneron Genetics Center; SHARE Consortium; SPARC IBD Network; UK IBD Genetics Consortium; Duerr RH, Franke A, Brant SR, Cho J, Weersma RK, Parkes M, Xavier RJ, Rivas MA, Rioux JD, McGovern DPB, Huang H, Anderson CA, Daly MJ. Large-scale sequencing identifies multiple genes and rare variants associated with Crohn's disease susceptibility. Nat Genet. 2022 Sep;54(9):1275-1283. doi: 10.1038/s41588-022-01156-2. Epub 2022 Aug 29. PMID: 36038634; PMCID: PMC9700438.
- Ricciuto A, Aardoom M, Orlanski-Meyer E, Navon D, Carman N, Aloi M, Bronsky J, Däbritz J, Dubinsky M, Hussey S, Lewindon P, Martín De Carpi J, Navas-López VM, Orsi M, Ruemmele FM, Russell RK, Veres G, Walters TD, Wilson DC, Kaiser T, de Ridder L, Turner D, Griffiths AM; Pediatric Inflammatory Bowel Disease–Ahead Steering Committee. Predicting Outcomes in Pediatric Crohn's Disease for Management Optimization: Systematic Review and Consensus Statements From the Pediatric Inflammatory Bowel Disease-Ahead Program. Gastroenterology. 2021 Jan;160(1):403-436.e26. doi: 10.1053/j.gastro.2020.07.065. Epub 2020 Sep 23. PMID: 32979356.
- Kesler SR. Turner syndrome. Child Adolesc Psychiatr Clin N Am. 2007 Jul;16(3):709-22. doi: 10.1016/j.chc.2007.02.004. PMID: 17562588; PMCID: PMC2023872.
- Gatti S, Gelzoni G, Catassi GN, Catassi C. The Clinical Spectrum of Inflammatory Bowel Disease Associated With Specific Genetic Syndromes: Two Novel Pediatric Cases and a Systematic Review. Front Pediatr. 2021 Oct 26;9:742830. doi: 10.3389/fped.2021.742830. PMID: 34765575; PMCID: PMC8576358.
- Agrawal M, Spencer EA, Colombel JF, Ungaro RC. Approach to the Management of Recently Diagnosed Inflammatory Bowel Disease Patients: A User's Guide for Adult and Pediatric Gastroenterologists. Gastroenterology. 2021 Jul;161(1):47-65. doi: 10.1053/j.gastro.2021.04.063. Epub 2021 Apr 30. PMID: 33940007; PMCID: PMC8640961.
- Hayward PA, Satsangi J, Jewell DP. Inflammatory bowel disease and the X chromosome. QJM. 1996 Sep;89(9):713-8. doi: 10.1093/qjmed/89.9.713. PMID: 8917748
- Knudtzon J, Svane S. Turner's syndrome associated with chronic inflammatory bowel disease. A case report and review of the literature. Acta Med Scand. 1988;223(4):375-8. doi: 10.1111/j.0954-6820.1988.tb15887.x. PMID: 3369318.
- Tajima T, Oishi M, Nakae J, Satoh K, Okuhara K, Kanaya Y, et al. Turner syndrome with crohn disease. Clinical Pediatric Endocrinology. 2001;10(2):121–4. doi:10.1297/cpe.10.121